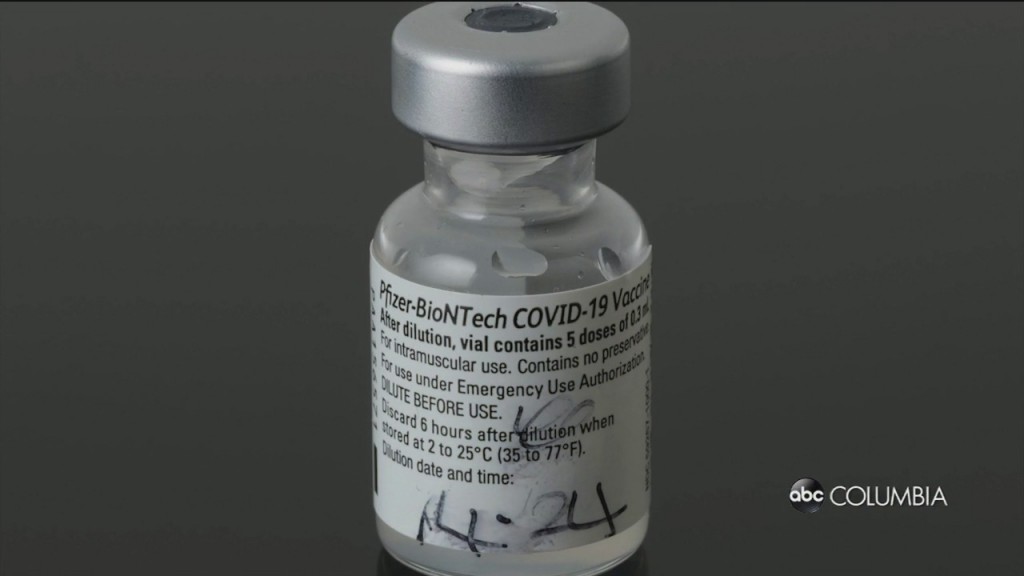
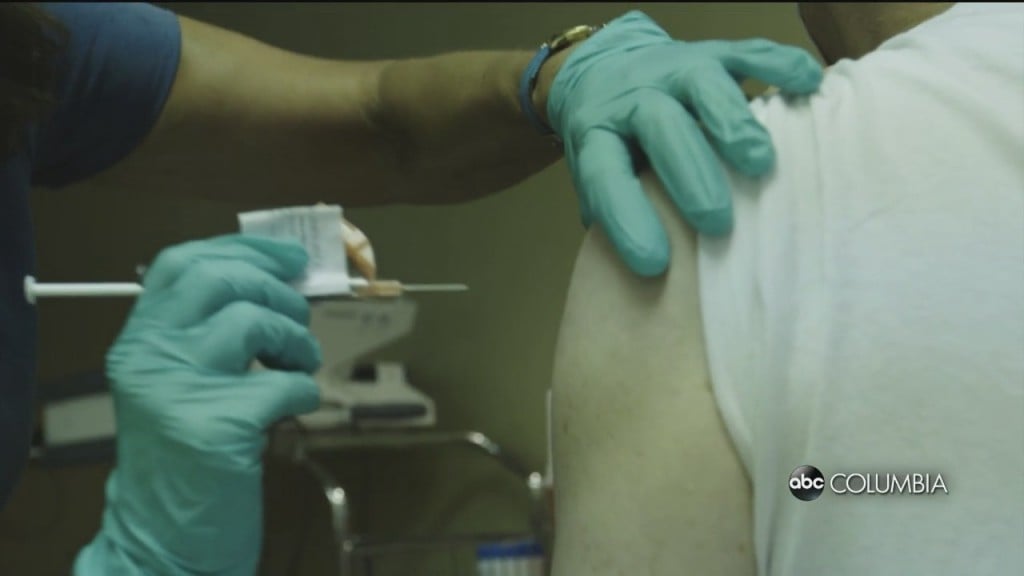
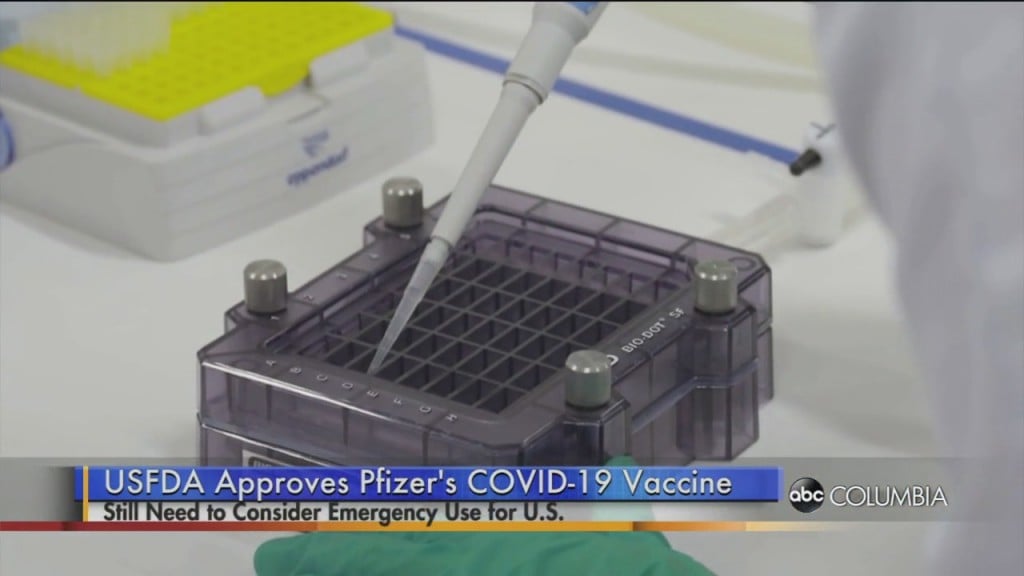
Pfizer, Moderna testing vaccine effectiveness against new strain of COVID-19 found in the U.K.
Pfizer and Moderna say they are testing to see if their vaccines work against the new COVID-19 strain which was found in the United Kingdom and other countries.